Surgical Robotics
We bring innovation in surgical robotics to life, FAST.
Cambridge Design Partnership is trusted by leading surgical robotics innovators to provide expert support across every stage of product development. Partnering closely with our clients’ teams, we help develop specifications and new system designs, create prototypes, scale existing complex systems, and resolve persistent development challenges. Our team finds the right path forward to solutions that combine technical robustness, commercial viability, and a user-centered approach.
Highly Specialized Surgical Robotics Services

Speed, Clarity, and Collaboration That Delivers
Speed with Confidence
We accelerate your timelines while reducing the risk of costly rework. Our staged, iterative approach delivers meaningful progress and actionable insight at every phase, from early architecture to pilot manufacture.
Flexibility Built In
Whether you need end-to-end support or targeted expertise, we adapt to your needs. From one-off sprint projects to full development programs, our collaborative model makes it easy to plug us in where and when we can add value.
Your IP. Your Success.
We don’t just work for you, we work with you. All IP and knowledge stays with you. We augment and empower your team, enhance your internal capabilities, and ensure seamless knowledge transfer at every stage.
Proven, Reliable Acceleration
Since 2019, we’ve developed multiple robotic surgery platforms, each reaching alpha prototype stage in under a year, to support pre-clinical trials and secure internal buy-in. That speed is no accident: it’s the result of having the right capabilities, capacity, and culture, all coordinated, all ready when needed.
Clarity Through Complexity
We bring clarity and structure to complex systems through requirements definition tailored to the current development stage, stakeholder alignment, and a multidisciplinary, systems-engineering mindset, ensuring focus, direction, and real momentum.
Clear Initial Stages With Confidence
Where some partners might require you to have specifications ready for them to work with, Cambridge Design Partnership often works with our clients through those challenging initial stages of a project. It’s all part of identifying the speediest, lowest-risk path to commercial viability.
How We Help You Accelerate
Integrated Surgical Robotics Expertise
Our cross-functional teams have delivered multiple complete Surgical Robotics systems since 2019, demonstrating speed, precision, and adaptability.

Pilot Manufacturing & Transfer
With our in-house 26,000 sq ft Pilot Production Centre, we enable early process development and smooth transition to manufacture.

Design Through Clinical Insight
Our integrated human factors team ensures that clinical insight is embedded early and continuously through all product development phases.
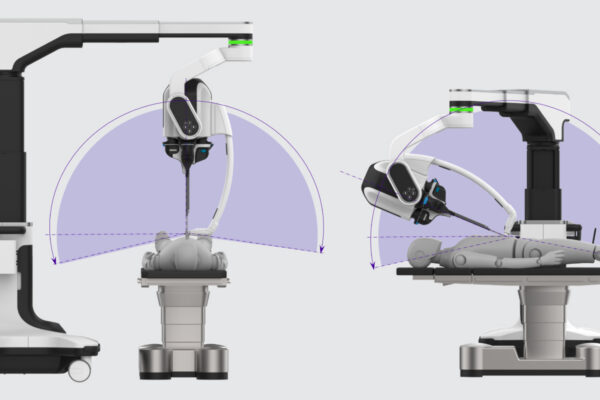
Built for Regulatory Success
We design to territory-specific medical device standards from day one, including IEC 60601-1 and others, supporting MDR submissions with zero major findings across project maturities.

Strategic US and UK Presence to Accelerate Innovation
Our UK and US offices position CDP to provide capability on-demand and facilitate seamless collaboration across all stages of Surgical Robotics development. These HQ locations gives us access to world-class technical talent, industry leading R&D partners, and leading clinical institutions, significantly enhancing our ability to deliver fast, scalable, and reliable results.
Our Raleigh North Carolina site supports rapid, high-quality onshoring of manufacturing. Coupled with our in-house 26,000 sq ft Pilot Production Centre at our Cambridge UK site we’re uniquely positioned to help you smoothly transition production stateside, efficiently navigate regulatory complexities, and reducing risks associated with offshoring. This local US capability, and deep regulatory and compliance expertise, ensures faster response times, increased supply-chain resilience, and accelerated time to market.

A Proven Partner to Deliver
End-to-End Thinking
We understand how today’s design decisions affect future manufacturing, regulation, and adoption. We think ahead on a component scale so that you don’t face unnecessary delays or rework later.
Total Capability, Ready to Scale:
Our 200-strong team includes engineers, scientists, designers, and regulatory experts across the UK and US. This breadth allows us to select project-specific teams with the right expertise, right away.
Close Collaboration
We embed seamlessly into your team; accelerating feedback, ensuring transparency, and building shared momentum. This integrated approach makes collaboration effortless and leads to stronger, more aligned outcomes.
Repeat-Proven Performance:
Our track record and presence in the surgical robotics industry speaks for itself. We’ve developed multiple robotic surgery systems, each in under a year, since 2019. This performance, and the trust it builds, means much of our work is from returning clients. They come back because we deliver: on time, to spec, and through collaboration that strengthens their internal capability as well as their product.

Positioned for Innovation
Located at the heart of innovation in Cambridge, UK, and Raleigh, North Carolina, our bases are strategically positioned to tap into the world’s leading research and development communities.

ISO-Certified Management System
To support our product development and prototype manufacturing services, CDP’s Quality Management System is certified to ISO 13485:2016 by Intertek Medical Notified Body and ISO 9001:2015 by Intertek Certification Ltd. We are also certified to ISO 27001:2022 by Intertek Certification Ltd to support the maintenance of the confidentiality, availability and integrity of our clients data.

Client-Centric Intellectual Property Approach
Our clients fully own all intellectual property we help create. We equip them with the tools and knowledge to enable lasting independence.

Ready to Accelerate?
Whether you’re leading an early-stage concept or refining a system for launch, CDP brings the clarity, capacity, and confidence you need to move forward. Contact us below and one of team will get back to you shortly.
Meet The Team
Capabilities
Featured Insights

How fast can you bring new Surgical Robotics tech to market? Know the 3Cs to rapid development.

CDP advances surgical robotic technology












