Insights from ESCMID 2025: trends and future in diagnostic testing
SUMMARY
This year’s ESCMID event reinforced that access to effective diagnostic tests must increase, and that long-promised technologies are finally close to delivering clinical value.
What makes your platform different?
One recurring message: speed, accuracy, and cost effectiveness are now expected. Differentiation lies in how well a platform fits real-world clinical settings, from training and workflow to data interpretation and system integration.
Takeaway for developers: Think beyond technical specs. Differentiation increasingly depends on usability, trust at scale, and measurable impact on care delivery and operational efficiency.
ESCMID 2025 marked a shift from technology-led innovation to outcome-led development. The key question is no longer “Can it be done?” but “Does it solve the right problem, in the right way, at the right scale?”
Headline Trends
- Antimicrobial resistance (AMR) and antibiotics use – increasing access to and uptake of diagnostic testing is key for effective interventions
AMR remains a major global health challenge. Antibiotic use continues to rise, especially in primary care, where most prescriptions are issued without diagnostic support. Addressing this requires a broader range of diagnostic systems. These must balance performance with affordability and usability, enabling appropriate treatment decisions in both hospital and community settings. - The role of syndromic testing is still being debated
While useful in hospital and acute care, syndromic panels have seen limited uptake in primary care due to cost and complexity. They support antimicrobial stewardship. But they must become more accessible and cost-effective to broaden adoption. - Genetic sequencing may bring a step change in clinical utility in diagnostics
Sequencing is moving closer to clinical use. It has the potential to reshape diagnostics. Developers must build workflows and systems that integrate sequencing seamlessly. They must interpret results clearly and deliver clinical value at scale. - ML, AI, and automation are maturing
AI tools are expanding from imaging to in-vitro diagnostics. They offer clinical augmentation value to tedious manual workflows, image interpretation, and data integration. The focus is shifting from innovation to implementation, embedding tools into workflows with trust, reproducibility, and regulatory alignment.
The annual congress of the European Society of Clinical Microbiology and Infectious Diseases (ESCMID) brings together clinicians, researchers, and industry leaders to explore the evolving landscape of diagnostics. This year’s event highlighted some key themes shaping the future of diagnostic impact and delivery.
AMR and Antibiotic Use: Access to Testing Is Critical
Antimicrobial resistance remains a central challenge. With an estimated 40–50 billion antibiotic doses taken daily and use projected to rise 50% by 2030, intervention is urgent. The vast majority of prescriptions occur in primary care. They are often without diagnostic support.
Improved access to diagnostics is key. Many current platforms focus on performance where demand already exists, but the biggest opportunity lies in reaching settings where no testing is currently available. Systems must support appropriate treatment decisions. They must balance speed, accuracy, and pathogen identification with usability, affordability, and integration into clinical workflows.
Syndromic Testing: Performance vs Cost
Multiplexed syndromic panels are established in hospitals and acute care, but uptake in primary care remains low. Their value in guiding antibiotic use is clear yet cost and complexity are barriers to broader use.
Developers must reduce system cost and complexity to reach more healthcare environments and fit into reimbursement frameworks. Technical capability alone is no longer enough. Integration, ease of use, and clinical decision support are now central to adoption.
Sequencing-based diagnostics are also beginning to compete with syndromic approaches, raising the bar for accessibility and performance in multiplexed testing.
Sequencing: Moving Toward Clinical Routine
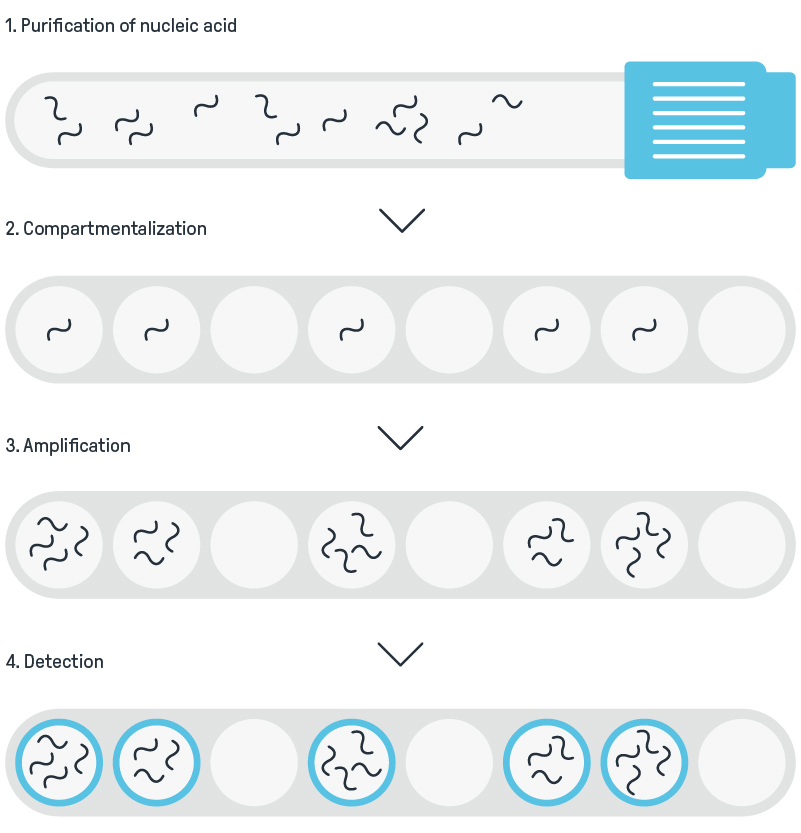
Genetic sequencing is rapidly approaching routine use in clinical diagnostics. With falling costs and expanding platform availability, it holds the potential to reshape infectious disease diagnostics, particularly in syndromic or multiplexed contexts.
At ESCMID, multiple case studies demonstrated the use of same-day metagenomic sequencing for pathogen identification in respiratory and bloodstream infections. Amplicon-based approaches were also discussed, particularly for rapid variant detection and resistance gene profiling. Broader sequencing methods, including microbiome analysis, may also play a role in future clinical applications.
However, sequencing workflows are not yet plug-and-play. Upstream processes, such as sample collection, DNA extraction, host depletion, and library preparation, remain technically demanding. Downstream, interpretation, bioinformatics pipelines, and clinically actionable reporting are major hurdles. While sequencing speed and cost are improving, the challenge now lies in integrating these steps into streamlined, automated, and interpretable systems that deliver value at the point of care.
Developers must ask strategic questions. Should initial diagnostic applications focus on relatively simple, targeted sequencing? For example, amplicon sequencing for variant detection, or on broader, hypothesis-free metagenomic approaches? Will sequencing be decentralized or remain concentrated in specialized hubs? How can systems present complex data in ways that support decision-making by non-specialist clinicians? The opportunity is significant. But realizing it will require systems that balance performance, usability, and clinical relevance.
ML, AI, and Automation: From Hype to Implementation
Artificial intelligence has already made an impact in diagnostic imaging, including MRI, CT, and ultrasound. At ESCMID, attention turned to the broader use of AI and machine learning in in-vitro diagnostics, with exciting potential across three main areas:
- Image analysis: AI is enabling rapid interpretation of high-resolution optical data in fields like digital pathology, haematology, and spectral imaging. These tools can increase diagnostic accuracy while reducing the burden on human reviewers.
- Multi-marker data interpretation: AI models are increasingly used to integrate complex biomarker datasets, where the combined signal across markers yields diagnostic insight. Some platforms already use deterministic models; the shift toward machine learning promises greater flexibility and performance, particularly as training datasets grow.
- Workflow optimization and automation: AI is being applied to streamline laboratory operations, reducing hands-on time, standardizing results, and minimizing error rates. This is especially valuable in high-throughput or resource-constrained settings.
Several ESCMID sessions showed AI being used to support antimicrobial resistance prediction, improve taxonomic resolution, and aid diagnostic decision-making. Importantly, the field is moving from exploratory development to real-world deployment. The emphasis is now on clinical validation, regulatory clarity, reproducibility, and integration with existing systems.
For developers, success will depend on more than accuracy. Trust, interpretability, and usability are emerging as key differentiators. Tools that embed smoothly into clinical workflows, minimize training requirements, and deliver repeatable, high-confidence outputs will define the next wave of AI in diagnostics.
Receive further news from Diagnostics at CDP
Connect with CDP
Wondering what these trends mean for your next diagnostic system? A quick conversation could help you shape a clearer path from concept to clinical impact. Get in touch with one of our team:
Dan Haworth, Head of Diagnostics
dan.haworth@cambridge-design.com
James Blakemore, Senior Insight and Strategy Consultant
james.blakemore@cambridge-design.com
Leigh Shelford, Consultant Physicist
leigh.shelford@cambridge-design.com