Reducing the carbon footprint and plastic waste of LFTs: Evidence-based opportunities
Billions of lateral flow tests have been used worldwide during the COVID-19 pandemic – over two billion have been provided in the UK alone. Debate has raged on social media about why the tests need to use so much single-use plastic and how they could be made more ‘sustainable’. The test strip caseworks is a particular source of dismay – why so much plastic to house such a tiny test strip?
With the UK government ending the free distribution of lateral flow tests for the general public – citing a transition from emergency response to longer-term management of the pandemic – now is the ideal time to look more closely at the sustainability of these lateral flow tests, and to seek the data to demystify some of the emotional assumptions being made.
Familiarity with lateral flow testing has certainly increased, as has confidence in their clinical performance. It’s expected that lateral flow devices will be more present in our daily lives post-pandemic – not just for COVID-19 and pregnancy testing but to diagnose diseases such as seasonal influenza and sexually transmitted infections – all from the comfort of the home.
We’ve carried out a high-level assessment to quantify the approximate environmental impact of lateral flow tests and identify evidence-based suggestions for improving their environmental sustainability.
Why do COVID-19 lateral flow tests contain lots of single-use plastic in the first place?
The emergence of COVID-19 was a global emergency, and vast quantities of lateral flow tests were needed urgently. Once developers could produce the right immunoassay chemistry to detect the virus (SARS-CoV-2), it required implementation in a low-cost, low-risk device, that has a mature supply chain – with proven, readily available materials that wouldn’t compromise analytical or clinical performance.
This meant using existing plastic casework designs to retain and protect the nitrocellulose test strip. Plastic is robust, low cost, lightweight, easy to transport, and easily printed for QR codes and LOT numbers. Critically, it’s a consistent material proven for the highest volume manufacturing and won’t interfere with the immunoassay chemistry.
From a performance, cost, and manufacturing perspective, redesigning the product with new materials would have been high risk. Material changes may also have needed significant R&D costs, new capital equipment as well as additional cost and effort needed to demonstrate equivalence and achieve regulatory approval – risking the ability to provide sufficient numbers of high-quality tests, at speed during the pandemic.
Our results: The sustainability of lateral flow tests
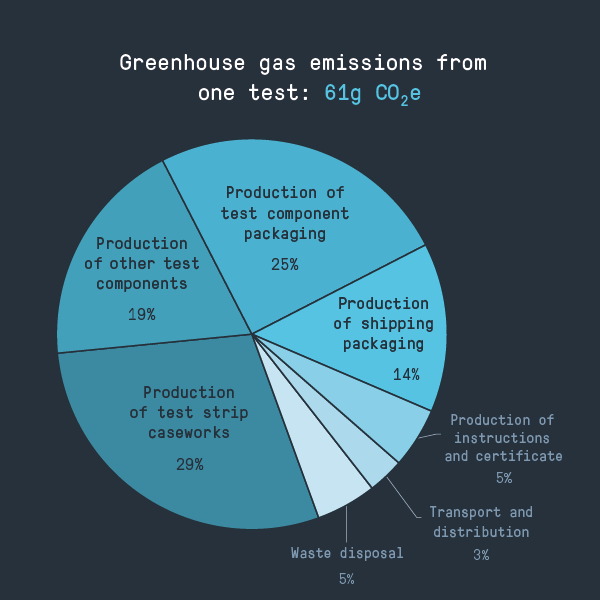
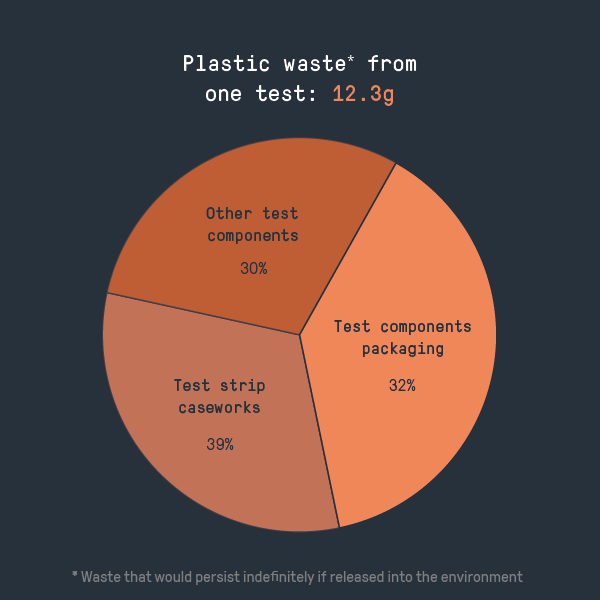
But how serious an environmental impact do these tests have? To find out, we broke down a test into its constituent components and weighed them to calculate the approximate environmental impact, using standard emissions factors to calculate the carbon footprint of a single test.

We focused on carbon footprint (the carbon dioxide and other greenhouse gases emitted during manufacture, transport, and disposal of the tests) and plastic waste (waste that would persist indefinitely if released into the environment) – the two issues that have attracted the most attention around lateral flow tests. A more comprehensive study should consider a broader range of environmental impacts, for example, the use of scarce resources and emission of other pollutants to avoid unintended consequences of any product changes.
Our results reveal:
- The components needed to conduct the test account for around half of the carbon footprint and around two-thirds of the plastic waste. Packaging makes up most of the rest – as is often the case, a surprisingly high proportion of the total environmental impact
- The test strip caseworks, which attracts the most comment online, is responsible for around 30% of the carbon footprint and 40% of the plastic waste. While it’s the most significant single contributor to the environmental impacts we evaluated, the large number of other small parts is also significant. Focusing on the caseworks therefore might not be the best strategy for improving the sustainability of the tests overall.
Lateral flow tests a minor piece of UK healthcare’s environmental impact
To put these numbers into context, we can compare the environmental impact of the two billion COVID-19 lateral flow tests distributed in the UK with the UK healthcare system’s overall environmental impact. We estimate the UK’s lateral flow tests have a carbon footprint equivalent to around 0.5% of the total NHS carbon footprint. This isn’t a trivial amount, but it’s also not the largest single contributor to the impact of the UK health system.
It’s also worth considering the positive environmental impact of a user-administered test on the health system. Conducting a test at home can eliminate the need for an individual to visit a test site, GP’s surgery, or hospital (assuming the clinical performance of the lateral flow test is adequate). Based on estimates from the Sustainable Healthcare Coalition, one lateral flow test has around 5% of the carbon footprint of a single GP appointment and produces a similarly low percentage of non-degradable (plastic) waste.
And that’s before we consider travel. We estimate one lateral flow test has the same carbon footprint as driving 350 metres in an average UK car. So, if you’re driving yourself to a test site or GP surgery some distance away, at-home lateral flow tests compare even more favorably.
If a lateral flow test prevents an individual from transmitting COVID-19 to a vulnerable person, there’s a public health benefit – as well as an environmental benefit – to keeping people out of the hospital. We can all see the discarded waste from home tests, but the less visible impact from energy- and material-intensive medical interventions is often significantly higher.
These approximate figures demonstrate why building an evidence base is vital during product development targeting sustainability objectives – because the results can be unexpected and non-intuitive.
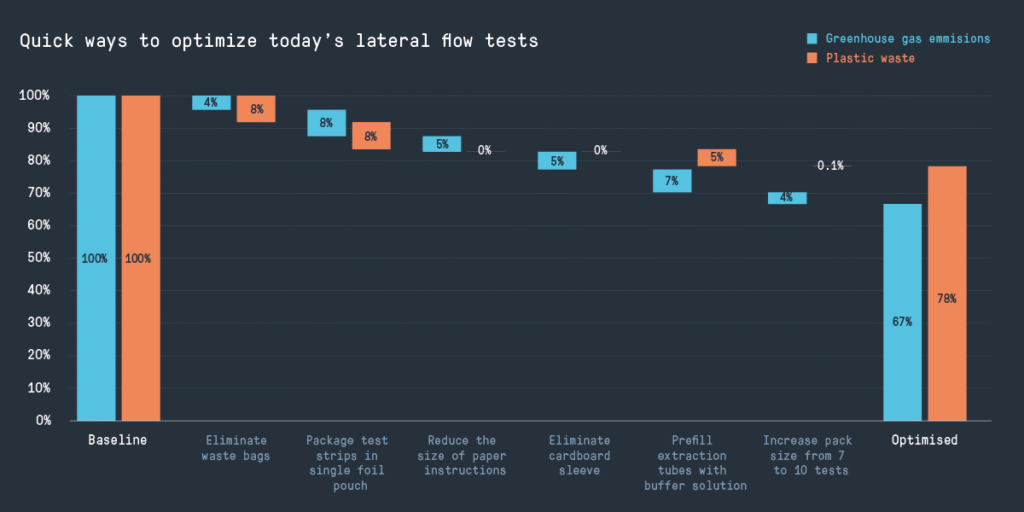
Quick ways to optimize today’s lateral flow tests
Just because waste from lateral flow tests might not be the most urgent sustainability issue for UK healthcare, that doesn’t mean we can’t and shouldn’t do something about it.
We used the ‘avoid/shift/improve’ model to find potential quick wins for lateral flow tests. These reduce the carbon footprint of each test by nearly a third and the plastic waste by almost a quarter – without impacting the fundamentals of how the test works.
They include:
- Eliminate waste bags. There’s a case for quickly isolating contaminated waste (even given COVID-19 also spreads from infected individuals through the air), but the bags account for around 5% of the carbon footprint of the test. It’s not clear how widely used they are in a domestic setting – there may be a risk-based justification for not including them in the test kit.
- Package all the test strips in a single foil pouch. Using a single re-sealable pouch to protect the tests from ambient humidity (rather than individually packing each test in a pouch with desiccant) is common in packs of lateral flow tests designed for use by healthcare professionals. However, once opened, the stability lifetime of the remaining tests is affected.
- Reduce the size of paper instructions. These are important for the effectiveness of the tests and are a regulatory requirement, but account for 5% of the carbon footprint of a test – could they be reduced in size?
- Eliminate the cardboard sleeve. This packaging isn’t essential to the safe and effective functioning of the test, and it seems likely that the functions it does provide could be achieved with less material.
- Prefill the extraction tubes with buffer solution. This is already done in some test kits, although manufacturers need to be conscious of moisture loss and the effect on shelf life. However, the separate plastic vial used in the test kit we studied accounts for around 5% of the carbon footprint and plastic waste.
- Increase the size of the pack from seven to ten tests. This would mean less package waste per individual test. Including ten tests in one pack instead of seven reduces the carbon footprint by around 5% (depending on how many other optimizations are done at the same time). Perhaps a pack of seven tests was originally designed to cover a week of daily testing – but is that how tests are being used in practice?
Redesign of the test strip caseworks
Looking to the longer-term gets us into product redesign – creating a new generation of the product with sustainability in mind. Doing this can take significant investment since, for medical devices, it’s likely to require new regulatory approval, which is a lengthy and costly process.
A popular idea circulating for lateral flow tests is to minimize the plastic test strip caseworks (without compromising the essential functions of providing a stable platform, and protecting the nitrocellulose test strip). It might be possible to halve the caseworks mass and reduce the overall carbon footprint and plastic waste by 15-20%. This would require significant investment in R&D, production tooling, and regulatory approval hoops to jump through – but could be worthwhile if future demand for tests stays high.
Longer-term options
If we consider that the world may require billions more lateral flow tests over the coming decade, a more comprehensive redesign becomes commercially viable. This could involve stripping the design back to the fundamental requirements for a lateral flow test – flowing a sample through the test strip in a way that is controlled and free from contamination. Current designs take advantage of established components to collect, buffer, and dose the sample – but, at this production volume, it may be worthwhile designing a system from the ground up that is optimized for cost, usability, performance, and sustainability.
Sustainability as a brand differentiator
It’s clear there’s scope to optimize lateral flow tests to reduce their environmental impact – and a systematic analysis reveals options beyond those that might jump out to someone when they use the tests. But it’s essential to put the impact of lateral flow tests in the context of the wider healthcare system, to focus resources where they can have the most environmental impact – and to recognize that, sometimes, the plastic waste people can see helps to avoid more serious, but less visible consequences.
On the other hand, while visible plastic waste from lateral flow tests may not be the most pressing environmental issue facing the healthcare industry, it highlights the growing influence consumer opinion is likely to have as diagnosis and treatment shift from hospitals to homes. And as lateral flow tests become (in the UK, at least) a product people buy with their own money, choosing from a range of options, there may be a competitive advantage for businesses that take note and optimize their products for sustainability.
References
- Prime Minister sets out plan for living with COVID [Internet]. GOV.UK. 2022 [cited 1 April 2022]. Available from: https://www.gov.uk/government/news/prime-minister-sets-out-plan-for-living-with-covid
- The Sustainable Healthcare Coalition. Care Pathways Calculator. [Internet]. Sustainable Healthcare Coalition. 2022 [cited 1 April 2022]. Available from: https://shcoalition.org/
Connect with CDP
For more information on reducing the environmental impact of lateral flow tests without compromising performance, contact Cambridge Design Partnership.