Life Sciences
Diagnostics & Life Science Tools
Our multidisciplinary team partners with you to expertly navigate the complexities of your diagnostic or life science system to accelerates you path to market.
Developing diagnostics and life science tools is challenging and complex. Success requires a multidisciplinary systems approach, combining a deep understanding of the core biological processes with specialist and full ranged engineering disciplines.
CDP’s experienced multidisciplinary team integrates diagnostics and life sciences expertise with our full range of product development capabilities. Our scientists and engineers excel in translating complex biological processes into robust product systems that deliver high performance and commercial impact. Whether developing low-cost consumables, complex instrumentation, or specialized analytical systems, we combine scientific insight with rigorous engineering to deliver solutions and accelerate innovation.
We have a rich track record in all stages of product development, from insights and early concept generation through to pilot manufacture and verification. Our ISO 13485-certified innovation process unites user needs with advanced technology and a deep understanding of the market dynamics. As a result, you’re always in safe hands.

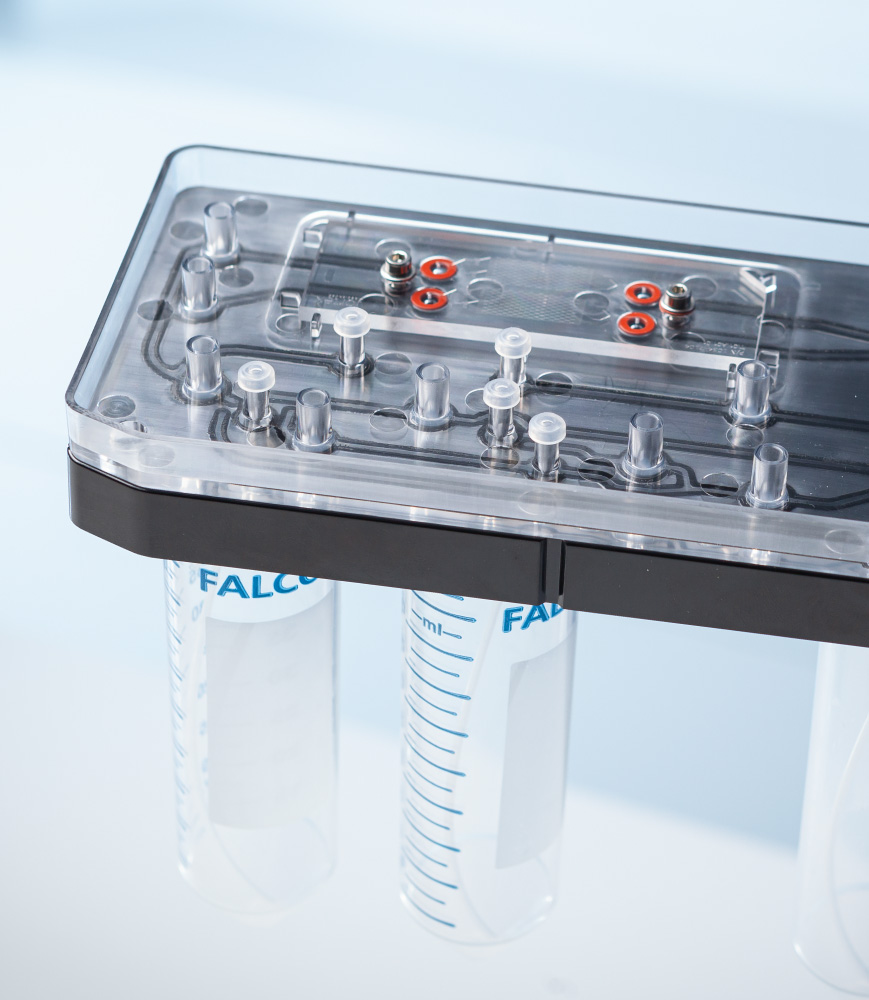


Life Science Tools
Developing laboratory and bioprocessing equipment requires a tight integration of biological insight with robust, full‑spectrum engineering. At CDP, we combine domain expertise with in‑house laboratories and prototyping facilities to design, build and test complex life-science tools, equipment and systems. Our multidisciplinary teams span engineering, physics and biology, enabling us to manage the critical interfaces – fluidics, detection, biology, usability and manufacturability – that define product performance.
Our experience includes:
- RUO and R&D systems design
- Applications for cell and gene therapy workflows
- Novel bioreactors and bioprocessing systems and associated control
- Considerations for GMP qualification
- Tools for genomics, sequencing and analytical workflows
- Cell biology (culture, processing and assay integration)
- Lab instruments, including liquid handling and robotics
Diagnostics
Through decades of experience in diagnostic product development, we’re practiced at implementing complex assays into compelling products for Lab, Point-of-Care and Home settings (across the regulatory spectrum, from RUO to IVDR). Our multidisciplinary team has deep understanding of the market and technological innovations to deliver competitive advantage, fast.
Our experience includes:
- Clinical diagnostics systems for high-throughput laboratories and decentralised point-of-care.
- Wearables and at-home health monitoring.
- Adapting lab-based R&D assay processes for cartridge and instrument automation.
- Bespoke cartridge and consumable development; concept generation, prototyping and scale-up.
- Custom development of core functional subsystems; multiplex optical detection, imaging, thermal control, fluidics
- Full system integration and optimisation
- Pilot manufacture and design verification

Developing Next-Gen Diagnostic Systems
Whether it’s unlocking the potential of a ground-breaking biomarker, innovating with new assay or detection technologies, or navigating the updates needed for your system to compete with newer devices, the path ahead is undoubtedly complex.
That’s why we’ve distilled the essence of successful diagnostic system development into this 10-minute guide – it’s your compass in the intricate world of diagnostics, highlighting critical considerations essential for turning ambitious ideas into tangible solutions.

Capabilities

Positioned for Innovation
Located at the heart of innovation in Cambridge, UK, and Raleigh, North Carolina, our bases are strategically positioned to tap into the world’s leading research and development communities.

ISO-Certified Management System
To support our product development and prototype manufacturing services, CDP’s Quality Management System is certified to ISO 13485:2016 by Intertek Medical Notified Body and ISO 9001:2015 by Intertek Certification Ltd. We are also certified to ISO 27001:2022 by Intertek Certification Ltd to support the maintenance of the confidentiality, availability and integrity of our clients data.

Client-Centric Intellectual Property Approach
Our clients fully own all intellectual property we help create. We equip them with the tools and knowledge to enable lasting independence.
Meet The Team
Featured Insights
How data and AI are changing bioprocessing – and why it’s needed

Insights from ESCMID 2025: trends and future in diagnostic testing

Five hurdles to digital health innovation in the UK (and how to overcome them)
FAQs
Explore our FAQs to find out more about how our Diagnostics team collaborate with you to expertly navigate the complexities of your diagnostic or life science system and accelerate your time to market.
Can CDP develop products that meet the complex needs of diagnostics and life science tools systems?
Yes, CDP’s multidisciplinary teams of scientists and engineers are experienced in translating biological processes to robust product systems that meet performance, cost and usability.
What stages of product development does CDP support?
We partner with clients over all stages, from idea generation and opportunity discovery right through to pilot manufacture. We structure projects to help our clients achieve their milestones on time and budget.
Does CDP develop products for regulated markets?
Yes, CDP’s design and development process is certified to ISO13485 and is compliant with the requirements of FDA and IVDR. We can support our clients in generating traceable requirements, risk management, V&V planning and generating documentation suitable for regulatory submission.
How does CDP manage scale-up and transition from prototype to final product?
CDP follows a risk-mitigation approach to integrating functionality and complexity while maintaining a clear path to the final product. We demonstrate the solutions to the highest risks early in the development process and avoid expensive and time-consuming design changes later on.
Does CDP develop assays and biological processes?
CDP does not lead assay development, but our biochemists and biologists can help advise on ways to improve the assay, for example, for robustness and cartridge implementation. Our experts can advise approaches to transfer from the lab bench to an automated product.
Can CDP help us to accelerate our development and to meet key milestones?
CDP’s in-house product development team enables rapid prototyping and development. With all capabilities in one place we are perfectly placed to run flexible and efficient projects that achieve results fast.
What types of organisations does CDP partner with?
CDP works with a wide range of clients at different stages, from world leading start-ups to large and established international organisations. We tailor projects to deliver the value our clients need to align with their development plan and milestones.















