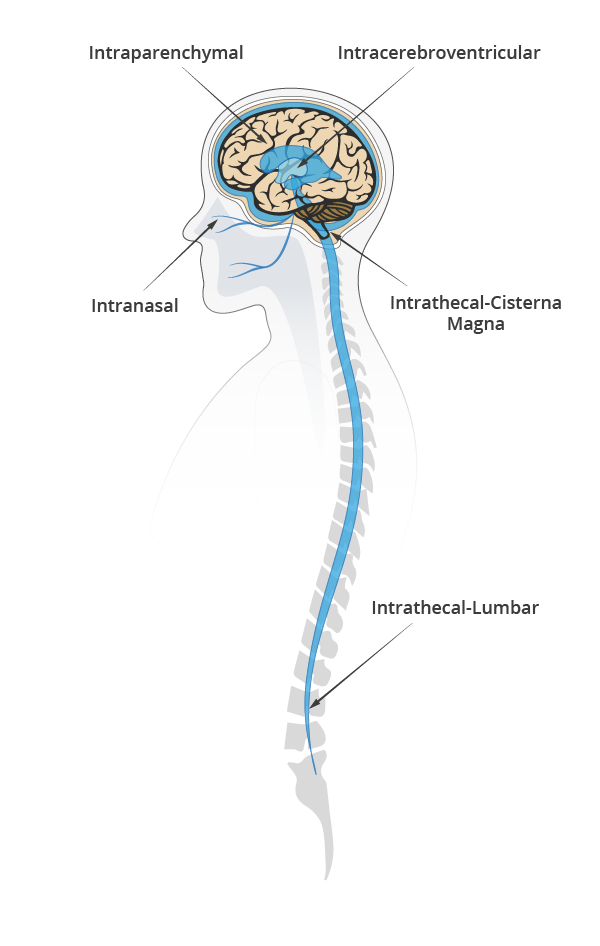
Epinephrine Injectors: Tackling the Prickly Problem for Kids
Improving Usability for Effective Emergency Treatment
In the United States, 5.6 million children – nearly 8% of the pediatric population – have food allergies, the leading cause of anaphylaxis in young children. More than 40% of these children have experienced a severe allergic reaction, and the incidence of food allergies is growing each year1.
Severe allergic reactions can rapidly become life-threatening, making the effectiveness of epinephrine injectors crucial. However, these devices often have significant design and usability issues, especially when used for pediatric cases.
The respiratory drug delivery landscape is undergoing change, driven by advances in technology, regulations, and evolving patient needs. While new trends open the door to innovation, they also bring about significant challenges that need to be addressed to ensure that respiratory care is optimal and accessible.
In this article, Lent Innovations’ Dr Annie Lent and Cambridge Design Partnership’s Steve Augustyn discuss:
Lent Innovations aims to set a new standard for pediatric anaphylaxis treatment by developing an epinephrine injector tailored to the specific needs of children, to reduce the risk of use errors and incorrect injection technique. The goal is to ensure that delivery of life-saving medication is easy for children and their caregivers.
Current Challenges in Epinephrine Device Use
A Clinician’s Frustrations with Current Epinephrine Injectors
Annie: Injection devices for epinephrine have always frustrated me, and they’ve always frustrated patients. They’re very confusing to use, leaving much room for error. They’re not appropriate for the treatment of anaphylaxis.
The two-handed process, where you have to take the cap off and then change hands to use the device, confuses many people. They often get mixed up about which end is up and which is down, leading to accidental thumb stabs and incorrect dosing, especially if it’s a caregiver administering the dose to a person suffering the allergic reaction. Many people also don’t know the force needed to inject properly, resulting in improper administration.
There’s been no focus on pediatrics either. We treat kids as little adults, not recognizing their specific needs and fears, which are distinct from adults. It’s intimidating for a child when someone approaches with a large injection device. The needle size is often too big for small children, causing pain and sometimes bone penetration. The devices are bulky, and many kids, especially teenagers, don’t want to carry one, let alone the two devices they are expected to carry. We need a better, less intimidating device.
Device Selection and Insurance Complications
Annie: When selecting from current devices to prescribe to patients, many options are available, but one major complication is insurance, as many companies won’t cover certain devices. Often, insurance companies or pharmacies substitute the prescribed device with an alternative that looks different and so is even more confusing. This means you train a patient on one type of device without knowing what they will actually receive from the pharmacy. One of our goals is to create the best device that is both accessible for and preferred by patients.

“One of our goals is to create the best device that is both accessible for and preferred by patients.”
Dr Annie Lent | Allergist and Immunologist, Founder of Lent Innovations
Innovation to Overcome Epinephrine Device Limitations
Device Reliability
Steve: One of the biggest disruptions we’ve seen in this market in recent years is the FDA’s focus on device reliability. This shift emerged from issues manufacturers faced when devices did not perform as reliably as required.
Unlike treating a chronic condition, where missing a dose might lead to symptom deterioration but allows for another opportunity to take the dose, life-threatening situations require immediate and reliable access to medication. Defining what constitutes reliability and proving it without astronomical test numbers is crucial. Considering the context of use is critical.
Emergency Use Situations
Steve: It’s the emergency use situation that sets these devices apart. If you were just looking to get an injection of a drug in a non-time-pressured manner for a chronic condition, there are fewer concerns. For example, you have much better control of the use environment, and the risk profile is much lower. With emergency use devices, you must consider not only the level of reliability needed but also the context of use. This context significantly impacts how you approach the design and functionality of these products.
Expert Insight and Iteration
Steve: Working with Lent Innovations, having Annie’s expert insight throughout the development process, has been a huge advantage. Immediate iteration and feedback have made a world of difference. Involving patients in our design process allows us to test, iterate, and refine continuously. With Annie’s access to a pool of patients, this approach becomes even more effective.
Focus on Transformative Design
Steve: Annie is an individual entrepreneur with finite resources. This means we must be super focused on delivering the greatest possible progress for the investment she has available. Our absolute focus has been on identifying where we can have the biggest transformative change to the design and iterating quickly. As soon as we have an idea, we de-risk it by moving quickly from 2D sketches to 3D models to ensure it works.
Importance of Understanding Combination Products
Steve: It’s so important to work with someone who understands combination products, the market, user requirements, and the context in which these companies operate. At Cambridge Design Partnership, we are fortunate to have a range of clients, from the biggest pharma companies in the world to disruptors like Annie with a real passion and vision for their products. When someone has an idea or has identified a problem that desperately needs a solution, it is much better to go to a company that truly understands the pharma market. Our drug delivery team has a deep knowledge of combination product development. They understand the regulatory and commercial constraints of working in this market and support our clients with expert insight.

“Our absolute focus has been on identifying where we can have the biggest transformative change to the design and iterating quickly.”
Steve Augustyn | Deputy Head of Drug Delivery
Market Trends and Dynamics in Epinephrine Delivery
Market Disruptions
Steve: We see several companies are trying to innovate with respiratory or nasal delivery of epinephrine, building on the work done with Narcan for opioid overdose. While I understand the benefits of taking it prophylactically if you think you’ve been exposed to an allergen, relying on respiratory delivery when you’re having a reaction and your airways are closing up is risky. You won’t be able to get the forced inhalation needed with a dry powder inhaler. Similarly, achieving reliable nasal delivery can be problematic if someone’s nasal passages are blocked due to rhinitis or normal biological processes. While these methods are interesting and may have a place, I believe needle-based delivery of epinephrine is here to stay.
Challenges in Pediatric Administration
Annie: In the pediatric world, trying to administer a nasal steroid to a child is nearly impossible. Kids run, thrash, resist, making it extremely difficult. Adding an emergency situation to the mix only makes it worse.
Future Trends in Anaphylaxis Treatment
Annie: People are going to be much more focused on building a better product. If inhaled and nasal methods do not work for everyone, in all situations, this will motivate people to make improvements. I hope Lent Innovation is setting a trend.
Steve: There will definitely be some rationalization in the market. Instead of focusing purely on the lowest cost, there will be a greater emphasis on solving usability problems. Lent Innovations’ project is emblematic of what can be achieved when the user is placed at the heart of the product, rather than driving everything by minimum cost. While we still have economic targets, it’s important to consider the lost opportunity and the human and economic cost of not receiving the injection properly.
“Instead of focusing purely on the lowest cost, there will be a greater emphasis on solving usability problems.”
Steve Augustyn | Deputy Head of Drug Delivery
As we look to the future, there must be a focus on developing reliable and user-friendly devices designed specifically for pediatric patients. This approach enhances the effectiveness of emergency treatments and ensures that children and their caregivers can confidently manage anaphylactic emergencies. By continuing to involve experts and end-users in the development process, we can achieve significant advancements in the field and set new standards for pediatric care.
Connect with CDP
At Cambridge Design Partnership, our approach prioritizes user experience to address the needs of healthcare professionals and patients and ensure intuitive device operation and efficient drug delivery.
If you have any questions about the content of this article, please get in touch with Steve Augustyn, Deputy Head of Drug Delivery at Cambridge Design Partnership: steve.augustyn@cambridge-design.com