Ocular Drug Delivery: Eyes on the Future
Advancing Sustained Release to Ease the Burden of Acute Therapies
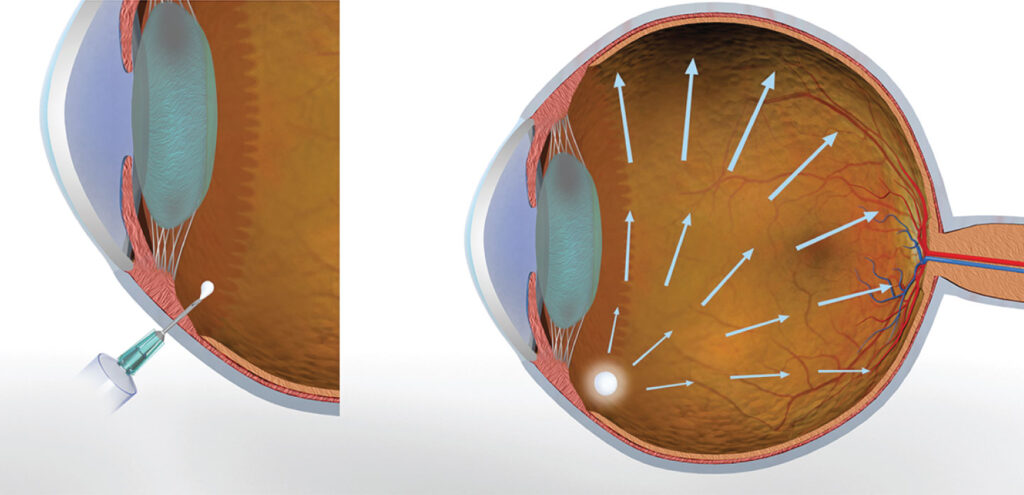
Complex drug products, delivered to specific tissue in the patient, can require a very innovative approach to delivery device development. Nowhere is this more important than for the treatment of ophthalmic diseases, where established delivery methods often require frequent hospital visits for injection into the eye by specialist practitioners, placing a heavy burden on the healthcare system – and the patient.
To alleviate this burden, pharma companies are developing sustained-release therapeutic assets, which aim to reduce the frequency of patient appointments. Achieving this requires a seamless integration of drug and device development.
In this article, Re-Vana Therapeutics‘ Patrick H O’Ruane and Cambridge Design Partnership’s Dariusz Zak and Fran Pencliffe discuss:
Re-Vana Therapeutics aims to transform ocular drug delivery with sustained-release large molecule biologics for a range of eye diseases. Their platform technologies, EyeLief®, EyeLief SD™, and OcuLief®, deliver biologics and small molecules. Their non-surgical approach targets a multi-billion-dollar market and offers expansion opportunities beyond ocular treatments.
The Rise of Targeted Combination Products
Shifting Perceptions in Drug Delivery
Patrick: Re-Vana has significant interest from multiple large pharmaceutical companies regarding our technology. There has been a fundamental shift in the industry where large pharmaceutical companies are keen to explore the advantage of targeted delivery as a way to extend the patent life of some assets and offer a more controlled method of delivering some of the more complex therapeutics.
Evolution of Combination Products
Dariusz: For a long time, devices such as pen injectors or auto-injectors were seen as necessary components to complete the expensive development of a drug that couldn’t be delivered orally. Drugs with a very narrow therapeutic window (such as insulin) have relied on highly accurate dosing for years. By applying this philosophy of highly accurate delivery to biological drugs, new therapeutic approaches open up. For example, the Re-Vana product requires both the delivery device and the drug to work together to achieve successful and targeted delivery for the sustained release of active pharmaceutical ingredients (APIs). We are witnessing a significant industry shift, emphasizing the need for highly capable teams to deliver these complex combination products effectively and efficiently within well-defined – and often truncated – timeframes. Accelerated development is necessary to align with investment needs and market demands.

“By applying the philosophy of highly accurate delivery to biological drugs, new therapeutic approaches open up.”
Dariusz Zak | Head of Applied Science
Enhancing Patient Care Through Sustained Release and User-Centered Design
Addressing the Burden of Acute Delivery
Patrick: There are numerous molecules aimed at treating retinal diseases, including wet age-related macular degeneration (AMD), dry AMD, geographic atrophy, diabetic retinopathy, and various genetic conditions. Despite the abundance of these drug assets, they all face a common challenge: they require patients to visit the doctor’s office for treatment every four to six weeks, placing a significant burden on the healthcare system. In the American healthcare system, there are approximately 2,500 retinal specialists, a number that is not increasing at the same pace as their caseload. Meanwhile, the number of patients diagnosed with retinal diseases is rising, and the population is aging. This creates a bandwidth issue for treating all these patients. Although clinical trials show promising results for many of the treatments, real-world data often falls short due to non-compliance by patients and shortage of healthcare provision. It is now widely accepted that what is needed is the sustained release of these therapeutic assets that will ease the burden on both patients and the healthcare system, whether they are small, medium, or large molecules.
Expanding Focus Beyond Ophthalmology
Patrick: We believe that our domain expertise in combination drug-device products, combined with our proprietary photo-crosslinked drug delivery technology, allows us to explore many opportunities outside of ophthalmology. For example, we are considering glucagon-like peptide-1 (GLP-1) agonists, which are seeing huge demand in the market. These patients typically need to self-administer injections weekly or bi-weekly. But what if they could self-administer every three or six months? We see substantial value in that.
User-Centered Design and Regulatory Challenges
Fran: Users have varying levels of training and different use environments, so products and devices must be able to compensate for these differences. This is why there is an increasing focus on user-centered design. Demonstrating that devices are safe and effective is a significant challenge, especially as we move towards targeted treatments that often require specialist application. Regulatory bodies require substantial evidence to demonstrate effectiveness, but the exact amount of evidence needed is often not clearly defined for novel treatments. This ambiguity can make it difficult to understand and meet regulatory requirements for new areas of technology.

“It is now widely accepted that what is needed is the sustained release of these therapeutic assets.”
Patrick H O’Ruane | Chief Operating Officer at Re-Vana Therapeutics
Driving Innovation Through Agility, Expertise, and Collaboration
De-risking the Development Process
Patrick: When developing a device to deliver our sustained release formulation, I wanted to de-risk the difficult parts before committing resources to development under design control. My goal was to answer the fundamental question: can this be done? We are attempting to set a world record by creating the largest implant ever performed through a 25-gauge needle, to be able to carry significantly higher drug loading and achieve truly targeted, sustained-release dosing. We needed to determine if this was feasible or just a pipe dream. The two main questions were: can it be done? The answer was yes. Can we build a minimally viable prototype flexible enough to work with multiple assets? Again, the answer was yes.
Assembling a Specialized Team
Patrick: We have a unique advantage in assembling a team that specializes in combination drug-device products who appreciate that to succeed, you must understand both drug and device development and integrate them seamlessly. The two components of the combination product cannot be developed in isolation. My top recommendation to investors is to ensure they have a team capable of handling both aspects. It doesn’t matter if you have the world’s greatest drug development expert, even a Nobel Prize winner; you need a team that can manage both drug and device development.
Understanding Real-World Needs
Patrick: The last thing you want is scientists or engineers developing an idea in isolation – you need to talk to the end users throughout the development. In our case, this involves clinicians, the people treating patients, explaining the real problems. Many companies solve problems that don’t need solving, and doctors dismiss these solutions because they don’t need or want them. Moreover, if you solve a problem for physicians, you must do so without complicating their workflow or increasing costs. It might sound harsh, but it doesn’t matter if the product is better for the patient if it interferes with the clinician’s workflow or is too expensive – they often won’t use it. We’ve built a strong Scientific Advisory Board of leading retinal practitioners. Sometimes, we present ideas to them, and they provide invaluable feedback. For instance, if we consider a new approach, they might advise against it, saying no one will use it. This immediate feedback is crucial. Small companies with good scientific advisors can move quickly in this respect.
Balancing Vision and Flexibility
Dariusz: From my perspective, working with startups, I observe that balancing the company vision for the product with constructive feedback from end users can be challenging. It’s crucial to recognize when to pivot. While having a strong vision is important, feedback might indicate that you are not solving the right problem or that there are usability or specification challenges. These issues could render the device or idea impractical. Being able to pivot is often difficult because you need to stay committed to your vision while also being open to change based on user feedback, but this flexibility allows your product to evolve into something truly needed – and used – in the market.
“It is now widely accepted that what is needed is the sustained release of these therapeutic assets.”
Patrick H O’Ruane | Chief Operating Officer at Re-Vana Therapeutics
By focusing on sustained release technologies and user-friendly devices, we can significantly reduce the burden on healthcare systems and improve patient outcomes. Embracing collaboration between industry experts and end-users will drive the development of groundbreaking solutions, setting new standards in the treatment of complex diseases and enhancing the overall quality of care.
Connect with CDP
At Cambridge Design Partnership, we have extensive experience with combination products. Our rigorous, science-based approach enables us to address complex design challenges, ensuring efficient and effective solutions.
For enquiries regarding this article, please contact:
Dariusz Zak, Head of Applied Science
dariusz.zak@cambridge-design.com
Frances Pencliffe, Consultant Healthcare Devices Engineer
frances.pencliffe@cambridge-design.com